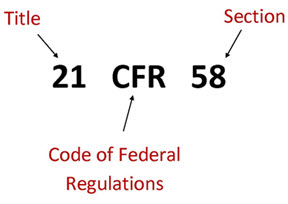
Book M2: 2022 Mini Pocket-Sized (3" x 5") Code of Federal Regulations – Clinical Research Resources, LLC

CFR 21, Parts 300 to 499, Food and Drugs, April 01, 2017 (Volume 5 of 9): Office of the Federal Register (CFR): 9781298709035: Amazon.com: Books

Food and Drug Administration Title 21 CFR Part 11 Technology Company Title 21 of the Code of Federal Regulations png download - 1300*1300 - Free Transparent Food And Drug Administration png Download. - CleanPNG / KissPNG

The Complete Code of Federal Regulations, Title 21, Food And Drugs, FDA Regulations, 2016 - Kindle edition by United States Government. Professional & Technical Kindle eBooks @ Amazon.com.

CODE OF FEDERAL REGULATIONS TITLE 21 Food And Drugs BUDGET EDITION 2018 PARTS 1-99: CFR TITLE 21: FEDERAL REGISTER, OFFICE OF THE: 9781731153425: Amazon.com: Books

Title 21 of the Code of Federal Regulations Title 21 CFR Part 11 Food and Drug Administration, others, text, brand, title 21 Cfr Part 11 png | PNGWing

National Hot Sauce Day and Food Regulations – Pic of the Week | In Custodia Legis: Law Librarians of Congress